Types of corrosion

Are you an existing customer or do you want to contact us in a different matter? We look forward to hearing from you and will get back to you as soon as possible.
Alternatively, you may want to contact directly one of our locations or our holding company.
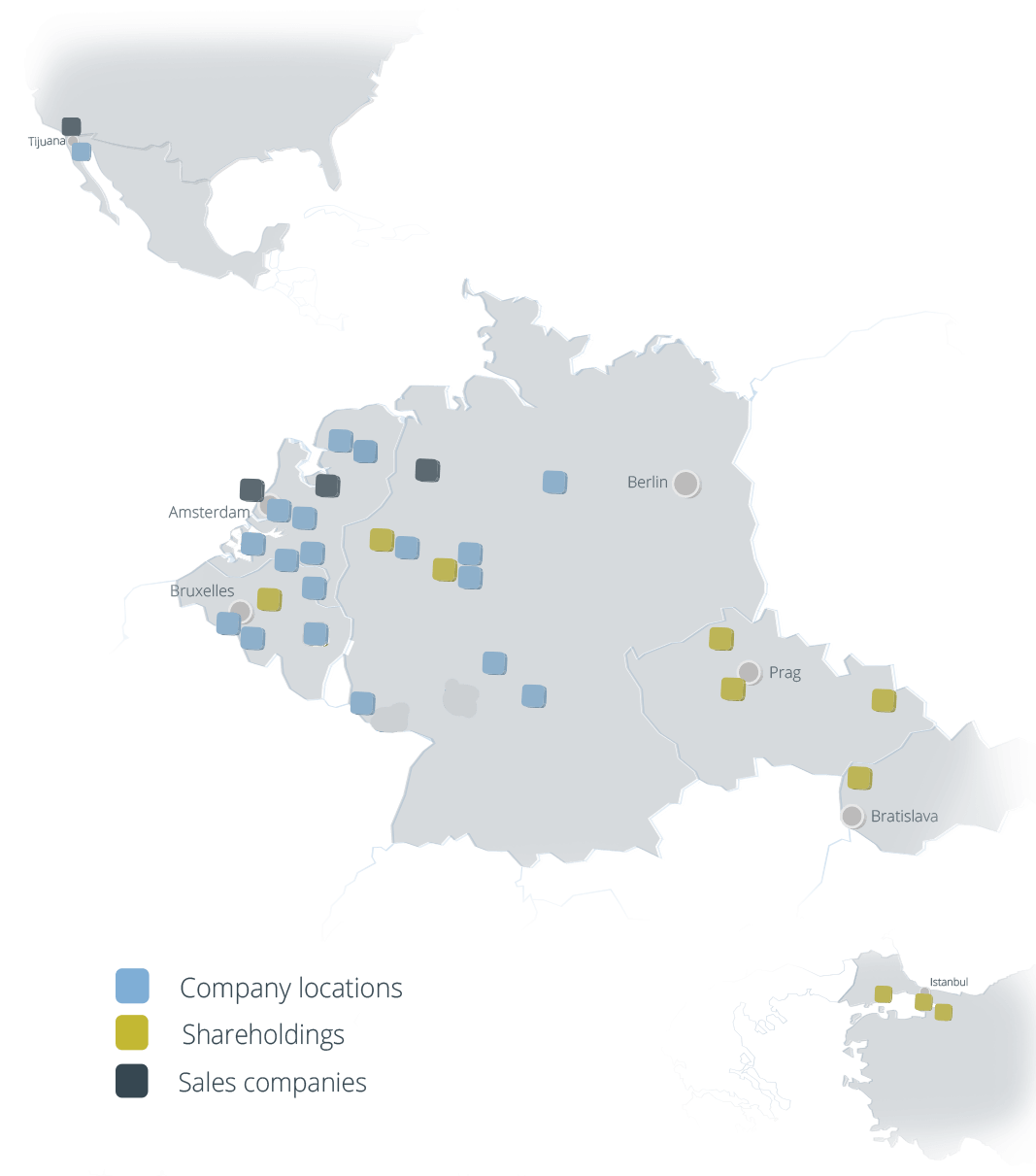
Locations
The Coatinc-Network
Netherlands
- Alblasserdam / NL
alblasserdam@coatinc.com - Amsterdam / NL
amsterdam@coatinc.com - Barneveld / NL
nederland@coatinc.com - De Meern / NL
demeern@coatinc.com - Groningen / NL
groningen@coatinc.com - Groningen – Pulverbeschichtung / NL
cgr.verkoop@coatinc.com - Mook / NL
mook@coatinc.com - Mook – PreGa / NL
prega.nl@coatinc.com - Roermond / NL
roermond@coatinc.com - Scherpenzeel / NL
anox@coatinc.com
For enquiries regarding marketing or press matters, please use the following contacts:
Marketing & Press
The Coatinc Company Holding GmbH
Hüttenstraße 45
57223 Kreuztal
Your contact person:
Anna-Maria Ademaj
marketing@coatinc.com
Holding
Headquarter
The Coatinc Company Holding GmbH
Carolinenglückstraße 6-10
44793 Bochum, Germany
Phone: +49 234 52905-0
Fax: +49 234 52905-15
Encyclopedia
Types of corrosion
Types of corrosion
Local corrosion attacks tend to occur because of existing defects when not caused by processing and construction errors. There is a difference between the material’s own intercrystalline corrosion (e.g. metal admixtures, which have a more precious potential than the basic metal); chemical corrosion which is foreign to the material (e.g. oxide layers, casting crust, annealing skin, blacksmith’s residue, mill scale on the material) and corrosion caused by the short-circuiting of metals with different electronegativity in the presence of water (electrolyte).
Differentiating types of corrosion:
The DIN standard DIN EN ISO 8044 defines a total of 37 different types of corrosion which are defined based on the location in which they occur:
- surface corrosion in which the surface is evenly damaged
- pitting corrosion in which the surface is unevenly severely damaged
- pitting, which damages only small parts of the surface
- crevice corrosion which occurs for example on welded seams
- hole pitting corrosion or pinprick corrosion in which small round pits quickly penetrate the material or workpiece
- dry corrosion in which very high temperatures cause iron oxide to form on the surface of the steel
- wet corrosion which occurs when mostly water (electrolyte) impacts on the component. This is the most common type of corrosion in the building industry.
- contact corrosion which occurs when different materials are connected with each other in the presence of an electrolyte.
By the way: corrosion on ferrous material is known as ‘rust’, corrosion on copper and Cu alloys is known as ‘patina’, and on zinc and Zn alloys it is ‘white rust’. And when high temperatures cause oxides to occur, this is known as ‘scale’.



