Surface technology

Are you an existing customer or do you want to contact us in a different matter? We look forward to hearing from you and will get back to you as soon as possible.
Alternatively, you may want to contact directly one of our locations or our holding company.
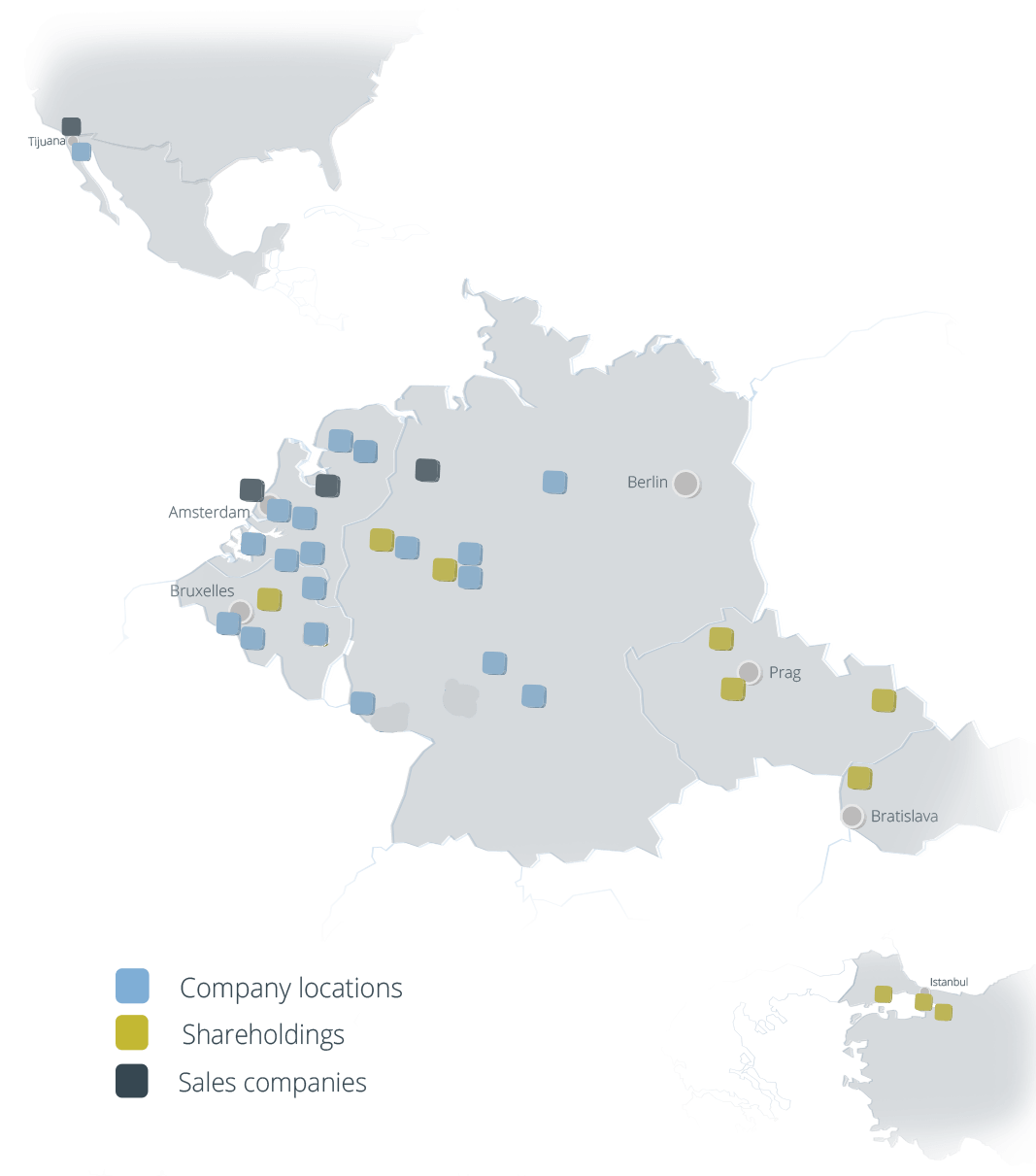
Locations
The Coatinc-Network
Netherlands
- Alblasserdam / NL
alblasserdam@coatinc.com - Amsterdam / NL
amsterdam@coatinc.com - Barneveld / NL
nederland@coatinc.com - De Meern / NL
demeern@coatinc.com - Groningen / NL
groningen@coatinc.com - Groningen – Pulverbeschichtung / NL
cgr.verkoop@coatinc.com - Mook / NL
mook@coatinc.com - Mook – PreGa / NL
prega.nl@coatinc.com - Roermond / NL
roermond@coatinc.com - Scherpenzeel / NL
anox@coatinc.com
For enquiries regarding marketing or press matters, please use the following contacts:
Marketing & Press
The Coatinc Company Holding GmbH
Hüttenstraße 45
57223 Kreuztal
Your contact person:
Anna-Maria Ademaj
marketing@coatinc.com
Holding
Headquarter
The Coatinc Company Holding GmbH
Carolinenglückstraße 6-10
44793 Bochum, Germany
Phone: +49 234 52905-0
Fax: +49 234 52905-15
Encyclopedia
Surface technology
Surface technology
Surface technology aims to make surfaces usable for specific application purposes through coating. Surfaces need to fulfil the requirements expected of them, such as wear and corrosion resistance, strength, optics, shine, colour, etc. This is where the type of layer and coating are crucial, as well as the materials.
Substrate properties
Substrate properties are described by their bonding states between the elementary components and their spatial structure and distribution (saturated bonding states). Bonding states (valances) are differentiated:
- main valences: ionic, covelent and metal bonding;
- semi-valences: e.g. electrostatic interaction, the basic properties are defined by the type of chemical bonding.
Characteristics of metal bonding
There is a strong force between the metal ions (+) and the ‘free’ electrons (also known as electron gas or electron gas cloud). The so-called metal grid occurs. This is distinguished by high resistance, good ductility, extremely good conductibility for heat and electricity as well as partially high melting and evaporation temperatures.
Each processing procedure leads to changes in structure and texture. Consequences of this are changes in the electrochemical potential, solidification/hardening or also changes to the surface profile for example changes to the electromechanical potential, right up to negative values and susceptibility to corrosion resulting from this. Mechanical tensions partially develop as a result of solidification/hardening of the surface zone which, for example, can lead to insufficient adherence between surface and coat in a coating.



